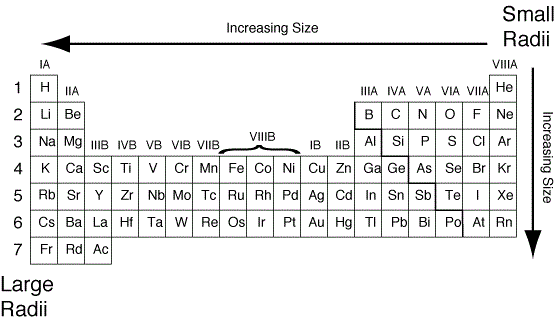 
It is a valuable alloying agent, as it contributes to the hardenability and toughness of quenched and tempered steels. It has a high elastic modulus, and only tungsten and tantalum, of the more readily available metals, have higher melting points. (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. Properties The metal is silvery white, very hard, but is softer and more ductile than tungsten. 1 which shows a plot of total electron density for all occupied orbitals for three noble gases as a function of their distance from the nucleus. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Atomic Radii Recall that the probability of finding an electron in the various available orbitals falls off slowly as the distance from the nucleus increases. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of iron-56 (atomic number: 26), the most common. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. Our own ab-initio investigations based on atomic relaxation in special quasi-random structures (SQS) show that the EFA correlates with the size distribution of the constituting metals: large. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. 
\( \newcommand\)) experienced by electrons in the outermost orbitals of the elements. Technical data for the element Iron in the Periodic Table Technical data for Iron Click any property name to see plots of that property for all the elements.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
